Fending off infectious disease
Toward a new strategy for effective oral vaccines
Briefs
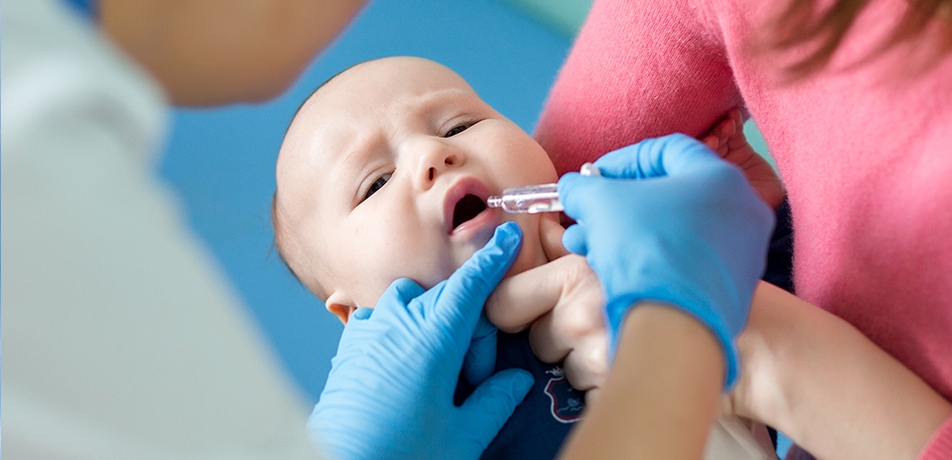
Since many major deadly infectious diseases—think smallpox, polio, diphtheria—have either been fully or mostly eradicated as a result of effective vaccines, the fact remains that many infectious diseases continue to lurk. Putting aside the public health challenge of overcoming defiance or fear around vaccinations—take the recent outbreak of measles among groups that forwent the MMR shot—the scientific toolbox for wiping out infectious disease is also still a work in progress.
At the level of basic research, Dr. Ziv Shulman, a member of the Weizmann Institute’s Department of Immunology is helping to elucidate the fundamental processes by which the human body develops immunity following vaccination. His findings—containing surprising news about how the immune response works differently in different parts of the body--may lead to more vaccinations that could be given orally, rather than through needles.
The harsh environment of the digestive tract—where gastric acid, bile, and enzymes work together to break down the food we eat—pose a major challenge for vaccine design, and indeed, most oral vaccines are not very effective. New research by Dr. Shulman has revealed that the body’s immune system also responds differently to vaccines delivered orally, as opposed to those delivered by needle injection. This discovery, based on the most detailed information ever achieved about how disease-targeting antibodies are produced in the gut, presents pharmaceutical companies with a new paradigm which may, in the future, contribute to the development of more—and more effective—oral vaccines.
Dr. Shulman is an expert in the adaptive immune response—the mechanism by which the body’s immune system recognizes and responds to harmful invaders. In adaptive immunity, different types of immune cells exchange molecular signals that allow them to differentiate into highly effective killers, capable of patrolling the body and eliminating specific pathogens.
In the recent study, published in the prestigious journal Nature Immunology, Dr. Shulman and Adi Biram—a PhD student in his lab— discovered that the production of pathogen-targeting antibodies in the gut follows a trajectory that is less efficient than this same process as it occurs in the body’s periphery.
In previous work, Dr. Shulman examined how, in the peripheral lymph nodes, selected B cells of the immune system—those cells most likely to secrete antibodies that bind to the specific target—are ushered into specialized training niches, where they undergo controlled mutations that make their protective immune response even more effective. An increase in the number of B cells having “high-affinity” antibodies generates a fighting force that provides long-term immunity against the pathogen in question.
In the current study, Dr. Shulman and Ms. Biram examined how B cells in the gut—as opposed to the lymph—produce their antibodies.
The researchers found that, in the gut, this process begins with the undifferentiated production of both cells that can produce both low-affinity and high-affinity antibodies. It is only in a second, further step that particularly “talented” B cells—the ones whose secreted antibodies have the best chance of binding to the invader—are ushered into a training niche, where further mutation polishes up these B cells’ pathogen-fighting potential.
scientists believe that their discovery—a differential “training programs” for B cells residing in the lymph and in the gut—may be rooted in the extreme complexity of the gut microbiome, which contains a huge complement of (mostly) helpful bacteria. In such an environment, it is more difficult for the body to positively identify a target, and then fine-tune the B-cell mediated immune response.
But whatever the reason behind its evolution, further study of this gut-specific immune response may yield new strategies for controlling how B cells in the gut respond to invaders—something that may eventually aid in the development of new or improved oral vaccines.
Dr. Ziv Shulman’s research is supported by the Morris Kahn Institute for Human Immunology; the Moross Integrated Cancer Center; the Sir Charles Clore Research Prize; the Rising Tide Foundation; the Azrieli Foundation; the Irma and Jacques Ber-Lehmsdorf Foundation; the Gerald O. Mann Charitable Foundation; and the David M. Polen Charitable Trust.

Dr. Ziv Shulman