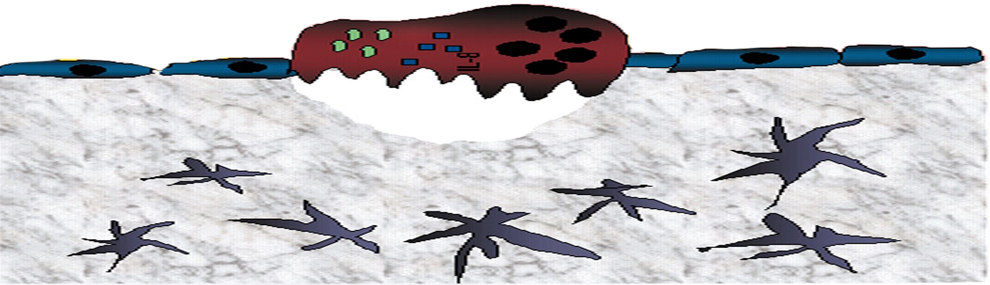
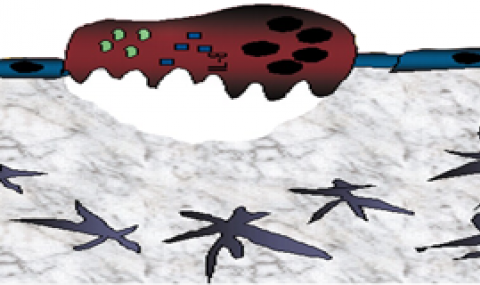
Hematopoietic stem and progenitor cells (HSPC) are mostly retained in the bone marrow (BM) in a ROSlow quiescent mode via adhesion interactions with stromal cells, while mature blood cells are generated on demand via ROShigh stem cell proliferation and differentiation.
The metabolic state of BM stromal cells and their cross-talk with quiescent and cycling HSPC is poorly understood. The bioactive lipid Sphingosine 1-Phosphate (S1P) is an important inducer of ROS in HSPC, increasing their motility (Golan et al, Blood 2012). We report that by increasing S1P levels in the murine BM, we also increased HSPC levels via augmented ROS production and cell cycling.
Unexpectedly, we discovered an opposite effect of S1P on BM stromal cells, which contained less mesenchymal stem and progenitor cells (MSPCs) and also lower ROS levels.
To further test the opposite metabolic relationship between BM HSPC and stromal cells, we utilized 3 repeated (once every 24h) G-CSF stimulations, which induce HSPC proliferation without mobilization. As expected, we found increased HSPC levels in the BM accompanied by augmented ROS levels.
Importantly, we found again the opposite effect between HSPC and BM stromal cells, i.e. reduced MSPC levels that were less cycling and contained lower levels of ROS. These results suggest an inverse metabolic state between BM HSPC and their stromal microenvironment.
While HSPC utilize oxidative phosphorylation and ROS generation during proliferation and differentiation, their stromal microenvironment share a low metabolic state, most probably due to anaerobic glycolysis.
Mimicking alarm situations by clinical G-CSF treatment blocks bone remodeling to allow HSPC development and mature blood replenishment. As expected, mice with reduced S1P levels due to lack of one of its biosynthetic enzymes (Sphingosine kinase 1), exhibit lower levels of BM HSPCs accompanied by reduced ROS levels.
More importantly, these mice share an augmented population of undifferentiated, primitive ROSlow, quiescent non-motile HSCs identified as EPCR+/LSK cells, which results in higher levels of long-term HSC repopulation in transplanted wild type mice (with normal S1P levels).
Concomitantly, S1P low mice have BM MSPCs with increased ROS levels and higher colony-forming unit fibroblasts (CFU-F) potential in vitro. We conclude that when HSPC are maintained quiescent due to a low energy metabolic state of anaerobic glycolysis, the BM stromal microenvironment utilizes a high energetic metabolism by oxidative phosphorylation and ROS generation.
Our data suggests that the dynamic, inverse metabolic state between HSPC and the stromal microenvironment is induced by energy transfer between the two populations. HSPC can eliminate excess ROS post 5-FU treatment, by transferring ROS to BM stromal cells via connexin43 (Cx43) gap-junctions (Taniguchi et al, PNAS 2012). In agreement, we found that mice treated with a broad inhibitor of connexin gap-junctions (CBX), induced BM HSPC proliferation via ROS elevation, concomitant with down regulation of ROS in MSPCs. Interestingly, in mice with low S1P levels, CBX inhibition of connexins did not elevate HSPC ROS levels or their numbers, implying that cell contact blockage is essential, but not sufficient to induce proliferation. To induce HSPC proliferation and differentiation, an activator of ROS, such as S1P, is also needed to initiate cell cycle entry and blockage of Cx43, further inducing CXCL12 secretion to the circulation and HSPC migration.
Our results imply that once ROS reaches a certain level in HSPC, it is scavenged by the stromal microenvironment to prevent exhaustion of the hematopoietic stem cell pool and in parallel to initiate new bone development and remodeling.
Altogether, we have discovered a dynamic, opposite metabolic state between BM HSPCs and their supporting stromal microenvironment during quiescence, proliferation and differentiation of the two populations, which are both ROS regulated on a one population at a time basis.
Thus, either blood cell production or bone development takes place at the expense of the other. This metabolic seesaw is regulated via ROS transfer from HSCs to their supportive microenvironment via Cx43 gap-junctions, which also regulates surface CXCL12 expression that is essential for stem cell quiescence.